
With recent advances in understanding the developmental biology of mammalian and non-mammalian hair cells a variety of strategies have emerged to restore lost hair cells are being developed. Developing effective therapies to restore hearing loss is challenging, owing to the limited regenerative capacity of the inner ear hair cells. Our findings suggest that the HNPC-BS slice co-culture isĪbstract: Sensory neural hearing loss and vestibular dysfunction have become the most common forms of sensory defects, affecting millions of people worldwide. HNPC showed the ability to extend neurites into host tissue. The results demonstrate that co-cultured HNPCs compared to monocultures (1) survive better, (2) distribute over a larger area, (3) to a larger extent and in a shorter time-frame form mature neuronal and glial phenotypes. HNPC aggregates (spheres) were deposited adjacent to or on top of the BS slices or as a monoculture (control). Here we characterized the survival, distribution, phenotypic differentiation, and integration capacity of HNPCs into the auditory circuitry in vitro. We recently developed a post-natal rodent in vitro auditory BS slice culture model including the CN and the central part of the AN for initial studies of candidate cells. A human neural precursor cell (HNPC) is an appropriate donor cell to investigate, as it can be isolated and expanded in vitro with maintained potential to form neurons and glia. 
Therefore, replacement of SGNs for restoring the AN to improve hearing function in patients who receive a cochlear implantation or have severe AN malfunctions is an attractive idea. The AN is composed of processes and axons of the bipolar spiral ganglion neurons (SGN), forming the connection between the hair cells in the inner ear cochlea and the cochlear nuclei (CN) in the brainstem (BS). These results should stimulate further research into the development of a cell-based therapy for deafness.Ĭongenital or acquired hearing loss is often associated with a progressive degeneration of the auditory nerve (AN) in the inner ear. Moreover, when transplanted into an auditory neuropathy model, otic neuroprogenitors engraft, differentiate and significantly improve auditory evoked response (ABR) thresholds. We obtained two types of otic progenitors able to differentiate in vitro into hair cell-like cells and auditory neurons that display expected electrophysiological properties. Here, we present a protocol to induce differentiation from human embryonic stem cells (hESCs) using signals involved in the initial specification of the otic placode. Using stem cells to recover the damaged sensory circuitry is a potential therapeutic strategy. 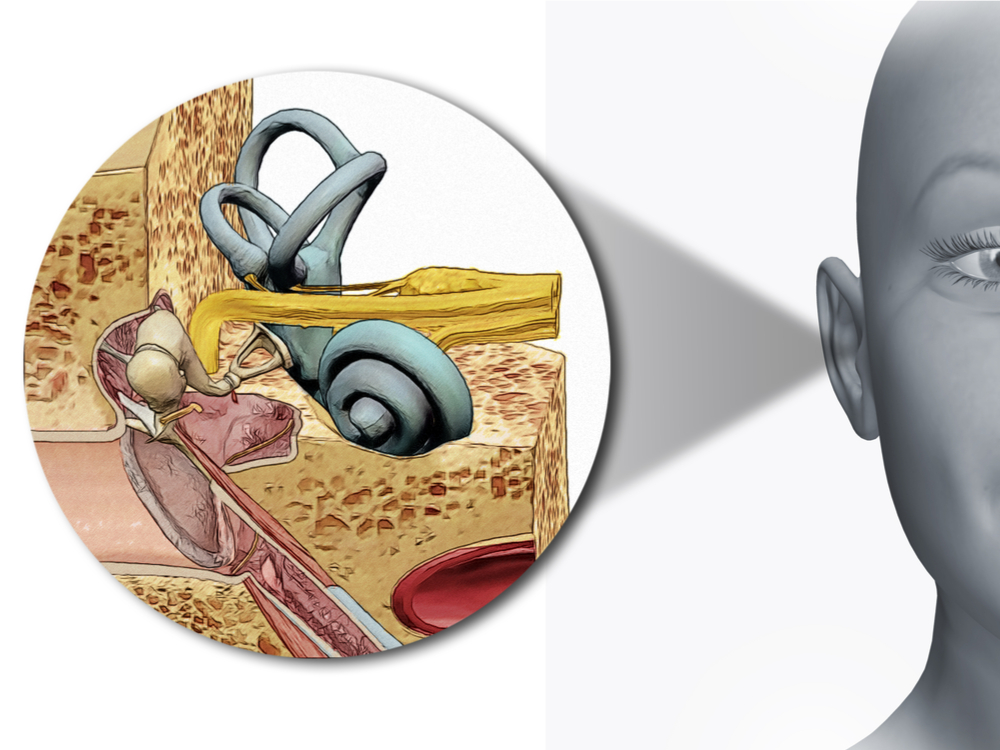
While the loss of hair cells can be circumvented partially by a cochlear implant, no routine treatment is available for sensory neuron loss since poor innervation limits the prospective performance of an implant 3. This condition, defined primarily by damage to the SGNs with relative preservation of the hair cells 1, is responsible for a substantial proportion of patients with hearing impairment 2. Of all the forms of deafness, auditory neuropathy is of a particular concern. Deafness is a condition with a high prevalence worldwide, produced primarily by the loss of the sensory hair cells and their associated spiral ganglion neurons (SGNs).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
